In situ oxidation transformation of trimetallic selenide to amorphous FeCo-oxyhydroxide by self-sacrificing MoSe2 for efficient water oxidation†
Abstract
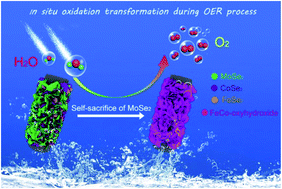
Transition metal chalcogenides have emerged as unique electrocatalysts for the oxygen evolution reaction (OER) during which they usually undergo an oxidation transformation into active oxides/(oxy)hydroxides. However, the transformation is so rapid that a high exposure of as-transformed (oxy)hydroxides cannot be achieved, thereby hindering the OER efficiency of the electrocatalyst. Herein, we report a simple self-sacrificing strategy to increase this exposure. A trimetallic selenide heterostructure (FeCoMo-Se) consisting of FeSe2, CoSe2 and MoSe2 is first one-step synthesized on a carbon cloth substrate. The heterostructure possesses a thin nanosheet morphology due to the support of MoSe2 nanosheets as a structural template. Under OER conditions, FeSe2 and CoSe2 are then in situ converted to FeCo-oxyhydroxide while retaining the nanosheet morphology of the heterostructure. Interestingly, MoSe2 is self-sacrificially dissolved and hence leaves considerable space to increase the exposure of FeCo-oxyhydroxide to the electrolyte. Such an advantageous nanostructure endows the FeCoMo-Se-transformed electrocatalyst with excellent OER performance in an alkaline medium, which is much higher than the non-MoSe2-containing selenide FeCo-Se. Density functional calculations demonstrate the favorable intermediate bindings in FeCo-oxyhydroxide. This novel self-sacrificing strategy opens up new avenues in the development of high-performance OER electrocatalysts with respect to their in situ oxidation transformation.


 Please wait while we load your content...
Please wait while we load your content...