Fe3O4/FeS2 heterostructures enable efficient oxygen evolution reaction†
Abstract
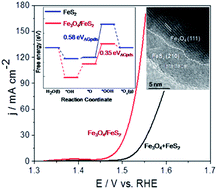
Although Fe has been occasionally recognized as the active site in the electrochemical oxygen evolution reaction (OER), monometallic Fe based catalysts still demonstrate insufficient OER activity in comparison to monometallic Ni and combined Fe–Ni materials. Herein, we report a highly active monometallic Fe catalyst by constructing Fe3O4/FeS2 heterostructures. Particularly, an in situ sulfuration route leads to a series of heterostructured Fe3O4/FeS2 materials which have tunable sulfuration degrees and variable contents of heterogeneous interfaces. At the OER conducted in an alkaline system, the acquired Fe3O4/FeS2-2.5 catalyst with the most abundant hetero-interfaces, exhibits superior performance by delivering ultralow overpotentials and good durabilities. Density functional theory computation reveals that charge redistribution at the interface region decreases the activation barrier for forming oxygen-containing intermediates which greatly accelerates the sluggish OER kinetics. This work clarifies that monometallic Fe based materials can deliver a high OER activity, and Fe should be the actual active site in this reaction. Besides, constructing heterogeneous structures with different anion species which received less attention before, may represent a promising strategy for designing highly active monometallic catalysts.


 Please wait while we load your content...
Please wait while we load your content...