Nitrogen electroreduction performance of transition metal dimers embedded into N-doped graphene: a theoretical prediction†
Abstract
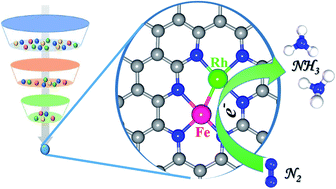
The electrocatalytic nitrogen reduction reaction (eNRR) under ambient conditions using sustainable energy sources is a fascinating approach for ammonia (NH3) production to replace the industrial Haber–Bosch process with enormous energy input and continuous CO2 emissions. However, the eNRR process remains a coveted goal due to the poor product selectivity, low activity, and stability of traditional catalysts. Inspired by recent experimental advances in dual-atom catalysts, herein, by means of comprehensive spin-polarized density functional theory (DFT) computations, the catalytic performance of various binary transition metals dispersed in nitrogen-doped porous graphene (NPG) for the eNRR was systematically evaluated. Our results revealed that the N atoms around vacancies in the substrate can strongly stabilize the metal dimers by forming metal–N bonds. Among FeFe, FeCo, FeNi, FeCu, FeRh, FeRu, FePd, MoFe, MoCo, MoCu, MoRu, and MoMo, the binary FeRh embedded in NPG was identified as the best catalyst due to its lowest limiting potential (−0.22 V) and high selectivity towards the eNRR. Our results further suggested that the good eNRR catalytic activity of the dispersed FeRh mainly originates from its optimal eNRR intermediates’ adsorption/desorption features, obvious suppressing effects on the hydrogen evolution reaction, and great promise for synthesis. Our work not only opens a new door to advance sustainable NH3 production by tailoring the electronic structures of binuclear metal atoms, but also provides deeper insight into the structure–performance relationships of dual sites for eNRR electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...