Unraveling the dissolution mechanism of platinum and silver electrodes during composite electrodeposition in a deep eutectic solvent†
Abstract
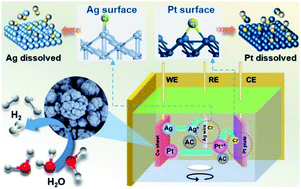
Composite electrodeposition, a versatile and convenient method, provides a strategy for the production of composite materials towards the high-performance hydrogen evolution reaction (HER). However, the insight into the dissolution behaviors of the electrodes during composite electrodeposition still requires further investigation. In this work, we report the dissolution mechanism of platinum (Pt) counter and silver (Ag) reference electrodes during Ni–activated carbon (AC) composite electrodeposition in a deep eutectic solvent. When AC particles were added into a deep eutectic solvent electrolytic cell with Pt as the counter electrode and Ag as the reference electrode, a three-dimensional Ni–AC–Ag–Pt composite with “cauliflower” morphology was produced, which exhibited great promising catalytic performance for the HER in the alkaline medium with a 48 mV overpotential at a current density of 10 mA cm−2 and a small Tafel slope of 35 mV dec−1. The improved HER performance is found to be mainly attributed to the dissolution–electrodeposition of Pt and Ag electrodes during the composite electrodeposition. Density functional theory calculations further demonstrate that Pt/Ag can react with the chlorine atom on their surfaces and the added AC particles have significant influences on the dissolution processes. These results suggest a new method for preparing Pt-based composites for the HER and also imply that the dissolution behaviors of Pt-counter and Ag-reference electrodes should be taken as general consideration for composite electrodeposition.


 Please wait while we load your content...
Please wait while we load your content...