Heterostructured CoP/MoO2 on Mo foil as high-efficiency electrocatalysts for the hydrogen evolution reaction in both acidic and alkaline media†
Abstract
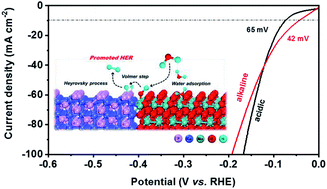
Heterostructured CoP nanoarray/MoO2 films on Mo foil (CoP–MoO2/MF) were synthesized through a low-temperature phosphorization process using pre-oxidized MF supported cobalt hydroxide-carbonate nanoneedles as the precursor. Such a CoP–MoO2/MF heterostructure exhibits superior HER performance with low overpotentials of 42 mV (alkaline) and 65 mV (acid) to achieve a current density of 10 mA cm−2, robust stability, large exchange current density and high turnover frequency. The experimental characterization and DFT calculations reveal that the presence of MoO2 leads to an inherent ability to adsorb water and cleave H–O bonds, favoring hydrogen transfer to accelerate the Volmer step. Heterointerfacial charge redistribution provides optimal hydrogen adsorption ability on adjacent P sites to boost the Heyrovsky step and, as a result, the hydrogen adsorption free energy (ΔGH*) can be reduced from 0.08 eV on CoP (011) and −0.78 eV on MoO2 (011) to 0.02 eV on the heterointerface of CoP–MoO2, which significantly improves the HER activity and accelerates the catalytic kinetics. This work highlights the roles of the heterointerface in electrocatalysis and provides an avenue to design electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...