Bimetal–MOF nanosheets as efficient bifunctional electrocatalysts for oxygen evolution and nitrogen reduction reaction†
Abstract
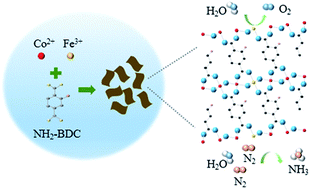
Metal–organic frameworks (MOFs) have emerged as promising electrocatalysts due to their controllability and diversity in functional species of metal centers and organic linkers. Herein, two-dimensional (2D) bimetal–MOFs (CoxFe–MOF) nanosheets with a thickness of ∼10 nm are synthesized and employed for OER and NRR in alkaline medium. By regulating the composition of CoxFe–MOF, the optimized Co3Fe–MOF can achieve an overpotential of 280 mV at a current density of 10 mA cm−2 with a small Tafel slope of 38 mV dec−1 in 1.0 M KOH electrolyte, outperforming the commercial RuO2 (351 mV at 10 mA cm−2, 99 mV dec−1). The enhanced OER performance can be attributed to the abundant active centers and the positive coupling effect between Co and Fe metal ions in the MOF. Additionally, the NRR performance of the obtained Co3Fe–MOF is evaluated on glassy carbon electrode to achieve real NH3 faradaic efficiencies (FE) and NH3 yield rates. The Co3Fe–MOF delivers a FE up to 25.64% with the corresponding NH3 yield rate of 8.79 μg h−1 mgcat−1 at −0.2 V versus the reversible hydrogen electrode (vs. RHE) in 0.1 M KOH electrolyte. The results indicate the potential application of 2D MOFs in NRR and provide the rational design of efficient electrocatalysts for coupling both OER and NRR in a full-cell configuration.


 Please wait while we load your content...
Please wait while we load your content...