Nanoporous bimetallic Zn/Fe–N–C for efficient oxygen reduction in acidic and alkaline media†
Abstract
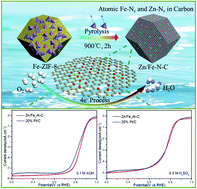
It remains a major challenge to develop a facile method to prepare non-noble metal electrocatalysts with high activity and durability to drive the sluggish oxygen reduction reaction (ORR) for proton exchange membrane fuel cells (PEMFCs). Herein, a highly efficient bimetallic Zn/Fe@N-doped hierarchical porous carbon (Zn/Fe–N–C) catalyst derived from ZIF-8 and hemin (host–guest) was firstly reported by one-step thermal treatment. Fe2+ spatially separated by using the hemin guest in the ZIF-8 framework and hydrocarbon-branched chains form rich Fe–Nx active sites, and the evaporation of Zn2+ generates a highly porous structure in the Zn/Fe–N–C catalysts. Benefitting from this unique structure and composition, the resulting Zn/Fe2–N–C catalyst exhibits excellent ORR activities in both 0.1 M KOH (onset potential, Eonset = 1.08 V, and half-wave potential, E1/2 = 0.86 V vs. RHE) and 0.5 M H2SO4 media (Eonset = 1.04 V and E1/2 = 0.81 V), which are even comparable to those of the commercial Pt/C catalyst, and ranks among the top reported electrocatalysts. In addition, it also has outstanding long-term durability and good methanol resistance, much better than those of Pt/C in both acidic and alkaline media, which makes it one of the best non-noble alternatives of Pt-based catalysts for ORR electrocatalysis. This work highlights the potential to rationally design and fabricate high-performance ORR catalysts for fuel cell applications.


 Please wait while we load your content...
Please wait while we load your content...