PdAg nanoparticles and aminopolymer confined within mesoporous hollow carbon spheres as an efficient catalyst for hydrogenation of CO2 to formate†
Abstract
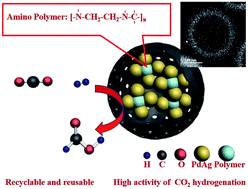
The hydrogenation of CO2 to formate is considered to be a reaction that can achieve the CO2-supported hydrogen energy cycle. However, the design of efficient heterogeneous catalysts possessing high activity for this reaction is challenging because of the high stability of CO2 and harsh reaction conditions. Herein, we design and synthesize ultrafine and well-dispersed PdAg nanoparticles (NPs) with an average diameter of 2.8 nm, which are dispersed uniformly within mesoporous hollow carbon spheres (MHCSs) containing a polymer (polyethylenediamine) with amino functional groups. Due to the limitation of the MHCS cavity spaces, the agglomeration of the alloy NPs could be effectively prevented, and finally the ultrafine PdAg alloy NPs and polymers are dispersed uniformly in MHCSs. The size of MHCSs is about 200–220 nm and the carbon shell is about 30–40 nm, which allows the dispersion of PdAg alloy NPs within MHCSs. Significantly, the PdAg alloy NPs in the vicinity of the aminopolymer dispersed in the hollow structured catalysts reveal enhanced catalytic activity and stability for CO2 hydrogenation to formate compared with control samples, showing a high turnover number (TON) of 2680 at 100 °C after 24 h. This synthetic strategy and the unique mechanism of core–shell catalysts for CO2 hydrogenation to formate will contribute to the CO2-mediated hydrogen energy cycle in the future.


 Please wait while we load your content...
Please wait while we load your content...