Covalent organic framework-based ultrathin crystalline porous film: manipulating uniformity of fluoride distribution for stabilizing lithium metal anode†
Abstract
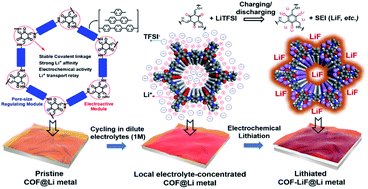
Lithium metal is regarded as the “Holy Grail” for rechargeable batteries yet it still suffers from low coulombic efficiency caused by its high reactivity toward the electrolyte. The so-formed solid-electrolyte interphases (SEIs) are ununiform and sluggish-ion-conducting further inducing severe dendrite growth. Herein, we report the in situ LiF-embedded covalent organic framework (COF) as a novel artificial lithium/electrolyte interphase. Briefly, an electrochemically active COF ultrathin film is synthesized by interfacial aldimine condensation, in which lithiophilic moieties reside in the high-surface-area COF film and can locally concentrate Li-salts from a dilute electrolyte. This promotes the electrochemical in situ formation of anion-derived LiF-rich SEI during the first cycling. Owing to the coaxially orientated microporous channels displayed in the COF, the as-formed LiF grains are confined in the micropores and evenly distributed throughout the COF matrix, thereby enhancing the mechanical strength of the SEI against dendrite growth as well as retaining flexibility to tolerate the anode volume change over cycling. Also, the Li+ diffusion pathway is highly ordered along the 1D pore walls of the COF so as to spatially homogenize Li+ flux. Therefore, the LiF-embedded COF interphase exhibits cooperative effectiveness to greatly stabilize the lithium metal anode. The full cell maintains its excellent performances with stable cycling and high efficiency in the close-to-practice conditions.


 Please wait while we load your content...
Please wait while we load your content...