Theoretical identification of layered MXene phase NaxTi4C2O4 as superb anodes for rechargeable sodium-ion batteries†
Abstract
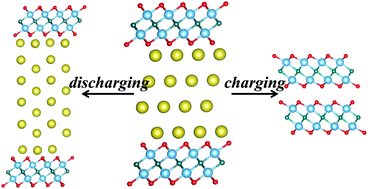
Rechargeable sodium-ion batteries (SIBs) have great potential as an alternative technology to substitute resource-limited lithium-ion batteries (LIBs). However, there is a greater safety concern in directly employing metallic sodium as the anode due to sodium being highly active chemically. It is also very challenging to develop SIB electrodes with high capacity as sodium ions are significantly larger and heavier than lithium ions. Here, in this work, we adopted a materials genome approach based on the density functional theory to screen stable phases through intercalating Na+ into a standard layered MXene phase of Ti4C2O4. It is highly appealing to note that each of the resultant series of MXene compounds of NaxTi4C2O4 (0 ≤ x ≤ 12) can maintain the layered structure of Ti4C2O4 through a fairly large extent of Na+ intercalation. The electrochemical potential vs. Na/Na+ is reversely correlated to the Na content: from 2.96 V and 2.40 V for NaxTi4C2O4 (1 ≤ x ≤ 2) down to 0.05 V for Na12Ti4C2O4. The total capacity for Na12Ti4C2O4 exceeds 579.00 mA h g−1, and this makes it a very promising high-capacity candidate as an anode for SIBs, since the structural integrity of the battery cells would be readily maintained over discharging/charging cycles. Besides, the O-terminated Ti4C2O4 layers are hydrophobic and oxygen-resistant; thus, the intercalated Na would be well protected in ambient processing conditions.


 Please wait while we load your content...
Please wait while we load your content...