Efficient gas and alcohol uptake and separation driven by two types of channels in a porous MOF: an experimental and theoretical investigation†
Abstract
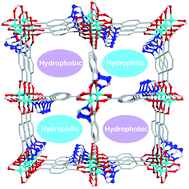
Utilizing the self-assembly strategy between the paddle-wheel Cu2(COO)4 cluster and a terephthalic acid ligand modified by a triazolyl group, namely H2tztp = 2-(1H-1,2,4-triazol-1-yl)terephthalic acid, a porous MOF, [Cu0.5(tztp)0.5]·0.5DMA (1), has been successfully designed and built. 1 is a three-dimensional self-interpenerated framework with a (3,6)-connected rtl topology, and contains hydrophilic and hydrophobic channels decorated with rich uncoordinated triazolyl N atoms and benzene rings from tztp ligands, respectively. The unique porous environment leads not only to high gas adsorption amounts of CO2, C2H6, C2H4 and C2H2 but also to a good separation ability of C2 hydrocarbons over CH4, CO2 over CH4, and C2H2 over CO2 in the MOF. The specific adsorption sites of CO2 and C2 hydrocarbons were explored by a molecular simulation. The breakthrough experiments demonstrated excellent dynamic separations for CO2 and CH4, C2 hydrocarbons and CH4 as well as C2H2 over CO2. The MOF also revealed excellent adsorption and separation ability for CH3OH over other bulkier alcohol molecules.


 Please wait while we load your content...
Please wait while we load your content...