Hollow PtCu nanorings with high performance for the methanol oxidation reaction and their enhanced durability by using trace Ir†
Abstract
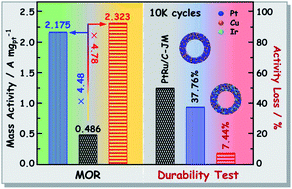
Platinum–copper (PtCu) alloy nanostructures represent an emerging class of electrocatalysts for the methanol oxidation reaction (MOR) in direct methanol fuel cells (DMFCs), but practical applications have been limited by catalytic activity and durability. In this study, we developed an efficient one-pot hydrothermal strategy to prepare unique PtCu alloy nanorings (NRs) with a highly open hollow structure. The formation process of the hollow NR structure includes the initial formation of pure solid Cu nanocrystals (NCs), the subsequent galvanic replacement reaction between Cu and Pt2+, and the co-deposition of Pt and Cu atoms at the edges. The r-Pt0.75Cu/C catalyst, that is, Pt0.75Cu NRs with an input Pt/Cu molar ratio of 0.75/1 supported on carbon black exhibited superior MOR performance, with a mass activity of 2.175 A mgPt−1 and a specific activity of 52.26 A m−2, which are 4.5- and 6.6-fold enhancements relative to those of commercial PtRu/C-JM, respectively. Impressively, the durability of Pt0.75Cu NRs for the MOR can be enhanced dramatically by doping with trace Ir. The mass activity loss of r-Pt0.75Ir0.05Cu/C, that is, Ir-doped Pt0.75Cu NRs supported on carbon black, was only 7.44%, much smaller than those of r-Pt0.75Cu/C (37.76%) and PtRu/C-JM (50.27%) after 10 000 CV cycles. This work provides a strategic design of efficient PtCu catalysts for the MOR.


 Please wait while we load your content...
Please wait while we load your content...