Unconventionally fabricating defect-rich NiO nanoparticles within ultrathin metal–organic framework nanosheets to enable high-output oxygen evolution†
Abstract
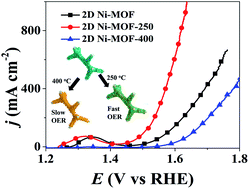
The high-temperature calcination of metal–organic frameworks (MOFs) often leads to a sharp collapse in the abundant pores inside the MOFs and a serious aggregation of metal sites, which are adverse to electrocatalysis performance. Here, a controllable calcination route was developed for the partial decomposition of ultrathin 2D Ni-based MOF (2D Ni-MOF) precursors to fabricate ultrafine NiO nanoparticles (NPs) within the ultrathin 2D Ni-MOF. In particular, 2D Ni-MOF precursors (thickness: ∼2 nm), for the first time, were rapidly synthesized via a microwave-assisted solvothermal method. The controllable calcination route effectively retained the ultrathin 2D porous nanostructure of the MOFs, and simultaneously enabled the formation of defect-rich ultrafine NiO NPs within the 2D Ni-MOF. Benefiting from the unique nanostructure (i.e., ultrathin 2D nanosheets) and highly active sites (i.e., defect-rich NiO NPs), the partially decomposed 2D Ni-MOF-250 exhibited excellent performance for oxygen evolution reaction (OER) with an overpotential of 250 mV at 50 mA cm−2 in 1 M KOH, outperforming those obtained from other reported nonprecious-metal-based electrocatalysts. More importantly, 2D Ni-MOF-250 could achieve the industry-related current density of 1000 mA cm−2 at a small overpotential of 410 mV, demonstrating its promising potential for use in practical applications. Therefore, the controllable calcination route may stand out as a facile yet robust route for smartly fabricating defect-rich metal oxides within MOFs toward efficient electrocatalysis.


 Please wait while we load your content...
Please wait while we load your content...