Engineering microporous ethane-trapping metal–organic frameworks for boosting ethane/ethylene separation†
Abstract
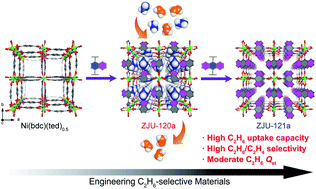
Realization of ethane-trapping materials for separating ethane (C2H6) from ethylene (C2H4) by adsorption, to potentially replace the energy-intensive cryogenic distillation technology, is of prime importance in the petrochemical industry. It is still very challenging to target C2H6-selective adsorbents with both high C2H6 capture capacity and gas selectivity. Herein, we report that a crystal engineering or reticular chemistry strategy enables the control of pore size and functionality in a family of isomorphic metal–organic frameworks (MOFs) for boosting the C2H6 uptake and selectivity simultaneously. By altering the carboxylic acid linker in Ni(bdc)(ted)0.5, we developed two novel isoreticular MOFs, Ni(ndc)(ted)0.5 and Ni(adc)(ted)0.5 (termed ZJU-120 and ZJU-121, respectively), in which the pore sizes and nonpolar aromatic rings can be finely engineered. We discover that activated ZJU-120a with the optimized pore size (4.4 Å) and aromatic rings exhibits both a very high C2H6 uptake (96 cm3 g−1 at 0.5 bar and 296 K) and C2H6/C2H4 selectivity (2.74), outperforming most of the C2H6-selective MOFs reported. Computational studies indicate that the suitable pore size and more nonpolar aromatic rings on the pore surfaces of ZJU-120a mainly contribute to its exceptional C2H6 uptake and selectivity. The breakthrough experiments demonstrate that ZJU-120a can efficiently separate C2H6 from 50/50 and 10/90C2H6/C2H4 mixtures under ambient conditions.


 Please wait while we load your content...
Please wait while we load your content...