Sulfide cluster vacancies inducing an electrochemical reversibility improvement of titanium disulfide electrode material†
Abstract
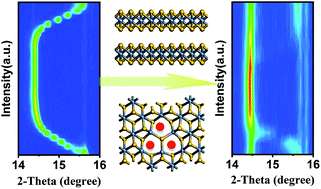
Promoting the reversible structural transformation and carrier migration dynamics simultaneously in a multiple ion-involved process is vital for sulfide electrode materials. Herein, TiS2 with a sulfide cluster vacancy (CV–TiS2−x) has been calculated and prepared to explore a novel strategy to improve the electrochemistry reversibility of sulfide electrodes. It is found that the cluster vacancy can serve as an effective control factor for the improvement of the Fermi level, and enhances the electrochemical stability of CV–TiS2−x in the liquid organic electrolyte system. Moreover, owing to the plentiful positively charged S-vacancies, the CV–TiS2−x samples exhibit an extended interplanar spacing and retain their structural integrity during the lithium ion insertion process. The integrated HRTEM, in situ XRD, XPS analysis and electrochemical study reveal that the cluster vacancy is not only responsible for an increase in reversible performance, but also boosting the anionic redox reaction of (S2)2− species. Therefore, the CV–TiS2−x electrode delivers a reversible capacity of 650 mA h g−1 for 300 cycles at a current density of 1C (220 mA g−1). Our study provides a novel strategy of introducing sulfur cluster vacancies to improve the electrochemical activity of TiS2, which can be applied to other sulfide electrodes.


 Please wait while we load your content...
Please wait while we load your content...