Understanding the structural phase transitions in lithium vanadium phosphate cathodes for lithium-ion batteries†
Abstract
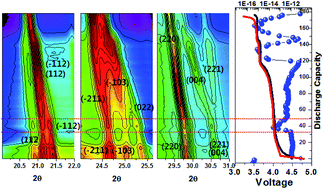
Developing high-energy lithium-ion batteries with long-term stability is critical for realizing sustainable energy applications; however, it remains highly challenging. Exploring multi-redox based electrode materials can help to achieve high capacity and high energy density in LIBs. Polyanion based monoclinic Li3V2(PO4)3 (LVP) cathodes have emerged as an interesting candidate for LIBs owing to their robust 3D structure, high theoretical capacity (∼197 mA h g−1), high working potential (>4.0 V), and high Li-ion diffusion coefficient. A vital challenge for the commercialization of LVP cathodes is their capacity fading behavior, originating when all three Li ions are removed (charging up to 4.8 V vs. Li/Li+). Understanding the phase transition and structural change during three Li-ion removal is indispensable to overcome the capacity fading in LVP cathodes. Herein, we explore for the first time the phase transition behavior responsible for severe capacity fading in LVP cathodes by combining synchrotron-based X-ray diffraction and GITT techniques. The study reveals the formation of a distorted Li-deficient Li1−xV2(PO4)3 phase upon third Li-ion removal which affects the consecutive Li-ion insertion, leading to a capacity loss. Furthermore, we reveal that the Li-ion insertion during discharge is a two-phase reaction, in contrast to the common understanding, which is a solid-solution reaction. This study emphasizes the importance of controlling distortion in polyanion frameworks to achieve high stability.


 Please wait while we load your content...
Please wait while we load your content...