Toward heat-tolerant potassium batteries based on pyrolyzed selenium disulfide/polyacrylonitrile positive electrode and gel polymer electrolyte†
Abstract
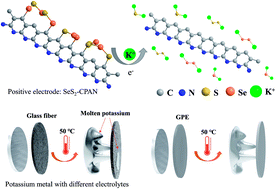
K–S batteries have attracted increasing research attention as promising energy storage systems. However, they suffer from slow reaction kinetics, which is attributed to the large radius of the potassium ion and insulating nature of sulfur. In addition, good heat tolerance of K–S batteries is crucial but not easy to realize due to the low melting point of potassium metal. Herein, we report a K–S battery that can be operated at both high and room temperatures with remarkable electrochemical performance using pyrolyzed selenium disulfide/polyacrylonitrile (SeS2–CPAN) as the positive electrode and a gel polymer as the electrolyte. SeS2–CPAN was prepared via calcination. Utilizing the advantages of both sulfur and selenium, the battery delivered a remarkable reversible capacity of 263 mA h g−1 (840.3 mA h gSeS2−1) at 100 mA g−1 and excellent rate performance (152.7 mA h g−1 at 2000 mA g−1) at room temperature. When the temperature was increased to 50 °C, the K//SeS2–CPAN cell with the liquid electrolyte and porous separator could not be cycled. However, with the gel polymer electrolyte, the cell delivered an initial reversible capacity of 304.9 and 229.6 mA h g−1 (974.1 and 733.5 mA h gSeS2−1) at 100 and 500 mA g−1, respectively, and it exhibited good cycle and rate performances as well. The gel polymer electrolyte mitigates the penetration of softened potassium and circumvents the short circuit, leading to remarkable electrochemical performance at a high temperature. Thus, this study demonstrates an effective and facile strategy to extend the application of potassium batteries, especially at high temperatures. In addition, the reaction mechanism and kinetics between SeS2–CPAN and potassium are discussed.


 Please wait while we load your content...
Please wait while we load your content...