Enhancement of Ni–(Y2O3)0.08(ZrO2)0.92 fuel electrode performance by infiltration of Ce0.8Gd0.2O2−δ nanoparticles†
Abstract
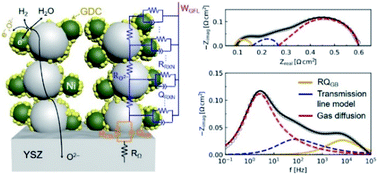
This paper addresses the use of Ce0.8Gd0.2O2−δ (GDC) infiltration into the Ni–(Y2O3)0.08(ZrO2)0.92 (YSZ) fuel electrode of solid oxide cells (SOCs) for improving their electrochemical performance in fuel cell and electrolysis operation. Although doped ceria infiltration into Ni–YSZ has recently been shown to improve the electrode performance and stability, the mechanisms defining how GDC impacts electrochemical characteristics are not fully delineated. Furthermore, the electrochemical characteristics have not yet been determined over the full range of conditions normally encountered in fuel cell and electrolysis operation. Here we present a study of both symmetric and full cells aimed at understanding the electrochemical mechanisms of GDC-modified Ni–YSZ over a wide range of fuel compositions and temperatures. Single-step GDC infiltration at an appropriate loading substantially reduced the polarization resistance of Ni–YSZ electrodes in electrolyte-supported cells, as measured using electrochemical impedance spectroscopy (EIS) at various temperatures (600–800 °C) in a range of H2O–H2 mixtures (3–90 vol% H2O). Fuel-electrode-supported cells had significant concentration polarization due to the thick Ni–YSZ supports. A distribution of relaxation times approach is used to develop a physically-based electrochemical model; the results show that GDC reduces the reaction resistance associated with three-phase boundaries, but also appears to improve oxygen transport in the electrode. Increasing the H2O fraction in the H2–H2O fuel mixture reduced both the three-phase boundary resistance and the gas diffusion resistance for Ni–YSZ; with GDC infiltration, the electrode resistance showed less variation with fuel composition. GDC infiltration improved the performance of fuel-electrode-supported full cells, which yielded a maximum power density of 2.28 W cm−2 in fuel cell mode and an electrolysis current density at 1.3 V of 2.22 A cm−2, both at 800 °C.


 Please wait while we load your content...
Please wait while we load your content...