An in situ structural study on the synthesis and decomposition of LiNiO2†
Abstract
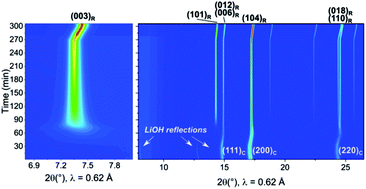
The electrification trend in the automotive industry is fueling research on positive electrode materials with high specific capacities. The nickel content in such layered oxide systems is continuously increasing, and so is the importance of LiNiO2 (LNO). Despite decades of research, LNO still exhibits properties, closely related to its instability, that require better understanding. One of these is the difficult solid-state synthesis that never seems to yield LNO samples of perfect stoichiometry. At present, improved experimental capabilities allow for investigating the synthesis process in unprecedented detail. Here, we leverage synchrotron X-ray diffraction, carried out in situ during calcination and decomposition of LNO at high temperature, to reveal the evolution of precursor materials during solid-state synthesis. We evaluate the effect of pre-annealing the hydroxide precursors’ mixture at low temperature, prior to the actual calcination at 700 °C. We then show that LNO formation is a structurally complex process, beginning from LNO seeds with a compressed rhombohedral unit cell (c/a < 4.9) within the rock salt framework. We identify a key aspect in the presence of Ni vacancies in Ni slabs, creating space for cation migration and allowing for the material's layering. We also investigate the decomposition of LNO, since it can be seen as the reverse process of synthesis. In fact, beginning already at 700 °C, it is in a way a byproduct of the synthesis. We correlate the change in stoichiometry with the unit cell volume of LNO and show how permanent damage is done to it by even a short time at too low O2 chemical potential. Taken together, this work aims at providing insights that may be of help in optimizing the synthesis of LNO while minimizing decomposition effects. Moreover, the same information can be seen as a starting point to further studies on Ni-rich (doped) compositions of practical interest.


 Please wait while we load your content...
Please wait while we load your content...