Preparation of biimidazole-based porous organic polymers for ultrahigh iodine capture and formation of liquid complexes with iodide/polyiodide ions†
Abstract
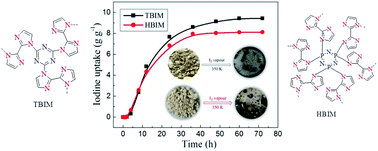
In the recent years, considerable progress has been made in iodine uptake—a radioactive emission process accompanying nuclear fission with porous organic polymers (POPs). However, all the resulting complexes of the cationic backbone of POPs and polyiodides are in the solid state. Here, we report, for the first time, liquid complexes that are formed from biimidazole-based POPs (TBIM and HBIM) and polyiodide ions. Interestingly, both TBIM and HBIM have excellent adsorption properties toward iodine with 9.43 and 8.11 g g−1, respectively. As far as we know, these are the highest adsorption values among all the POPs reported until now. Furthermore, TBIM and HBIM can sense both I2 and nitroaromatic compounds (NACs, such as picric acid/p-nitrophenol) via fluorescence quenching. In particular, TBIM has high fluorescence sensing performance toward iodine with the Stern–Volmer quenching constants (Ksv) of 1.16 × 104 L mol−1 and limit of detection (LOD) of 1.29 × 10−10 mol L−1.


 Please wait while we load your content...
Please wait while we load your content...