Exploring Ni(Mn1/3Cr2/3)2O4 spinel-based electrodes for solid oxide cells†
Abstract
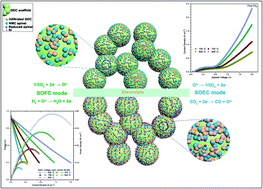
The alkaline earth element- and cobalt-free Ni(Mn1/3Cr2/3)2O4 (NMC) spinel is adopted to form composite electrodes with an oxygen ion conductive phase for symmetrical solid oxide cells and its applicability is successfully demonstrated in both solid oxide fuel cell (SOFC) and solid oxide electrolysis cell (SOEC) modes. In a H2 or CO–CO2 atmosphere, NMC decomposes to Ni and spinel, but would quickly recover to NMC in air or pure CO2. Both NMC and NMC–Gd0.1Ce0.9O2−δ (NMC–GDC) composite electrodes reveal a high redox resistance and fast recovery, which originated from the significant difference in Gibbs free energy when NMC is subjected to reducing and oxidizing atmospheres. In SOFC mode at 800 °C, the anode supported and symmetrical cells with the NMC–GDC electrode achieve peak power densities of 1293 and 416 mW cm−2, respectively. With an applied current density of 400 mA cm−2 at 750 °C in SOFC mode, the working voltage of the symmetrical cell with the NMC–GDC electrode showed no evident decline over 96 h. In SOFC mode and under pure CO2 in the cathode, the symmetrical cell working in SOEC mode showed a faradaic efficiency of over 96% at 700–850 °C and an electrolysis current density of 2320 mA cm−2 at 850 °C with an applied voltage of 2 V. Furthermore, the applied voltage decreased from 1.507 V to 1.407 V over a 72 h running time under a static current density of 800 mA cm−2 at 800 °C. The nickel exsolution in a reduction atmosphere and the abundant transition metal element centered octahedra ensure the multi-functionality of NMC. The NMC spinel-based electrode demonstrates an extraordinary redox resistance and running stability, and it is anticipated to play a pivotal role in the future exploration of novel and stable electrodes for solid oxide cells.


 Please wait while we load your content...
Please wait while we load your content...