Anodic engineering towards high-performance direct methanol fuel cells with non-precious-metal cathode catalysts†
Abstract
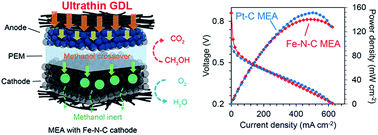
Direct methanol fuel cells (DMFCs) have drawn extensive interest for the past two decades both in scientific research and industrial engineering circles for their advantages of high energy density, environmental friendliness, and easy fuel handling. However, their excessively high costs, especially derived from the massive use of precious metal catalysts in both their anodes and cathodes, hamper the commercialization of this technology to the general public. Though the production of inexpensive catalysts of methanol oxidation remains challenging, non-precious-metal-based catalysts of the oxygen reduction reaction have seen considerable technical progress, yielding remarkable performance levels in hydrogen-fueled polymer electrolyte membrane fuel cells (PEMFCs). Due to the particularities of the electrochemical reactions and mass transport for methanol fuel in DMFC electrodes, highly active non-precious metal catalysts have not yielded sufficiently satisfactory single-cell performances for practical applications. In the current work, rather than exploring the cathodic designs with advanced electrode materials and structures, we estimated the mass transport of methanol and its effects on cathode performance, and then redesigned the anode architecture with ultrathin gas diffusion layers based on carbon nanotube composite materials. By using such an alternative strategy focused on anodic engineering to accelerate methanol transport combined with the use of a methanol-inert cathode, an ultrahigh cell performance, comparable to those of Pt–C-containing cathodes, was achieved even at a low methanol concentration. The peak power density obtained was 141 mW cm−2, a value among the highest obtained from DMFCs with non-precious-metal catalyst cathodes to the best of our knowledge. A wider avenue of DMFC technologies for practical applications might be opened with further development of this work.


 Please wait while we load your content...
Please wait while we load your content...