Constructing a tunable defect structure in TiO2 for photocatalytic nitrogen fixation†
Abstract
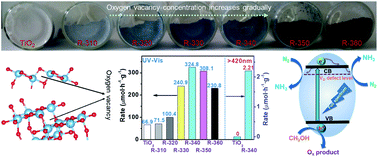
Photocatalytic systems capable of precisely regulating oxygen vacancy (OV) concentrations, which could help illuminate the effects of the OV concentration on N2 fixation activity, are still scarce. Here, we demonstrate that excessive OVs in TiO2, while increasing the adsorption activation capacity of N2 molecules, exhibit disappointing activity due to a decrease in charge separation efficiency. With optimized OV concentration, TiO2 can increase the charge separation efficiency 3-fold and show significant activation towards N2 molecules. The normalized N2 photofixation rate is 324.86 μmol h−1 g−1 (full spectrum) and the corresponding apparent quantum yield (AQY) under 365 nm illumination reaches 1.1%, which are relatively high levels compared to reports in the literature. The origin of this excellent activity is clearly attributable to the OV defect structures, which coordinate the charge separation efficiency and the dissociative adsorption capacity of N2. This work establishes the relationship between OV concentration and activity, and helps to construct a highly efficient nitrogen-fixing photocatalyst by optimizing the OV concentration.


 Please wait while we load your content...
Please wait while we load your content...