In situ decorated Ni2P nanocrystal co-catalysts on g-C3N4 for efficient and stable photocatalytic hydrogen evolution via a facile co-heating method†
Abstract
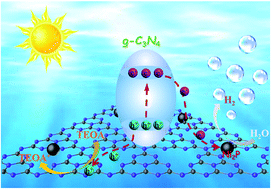
Very recently, transition metal phosphides (TMPs) have emerged as low-cost and robust co-catalysts for decorating graphitic carbon nitride (g-C3N4) for photocatalytic hydrogen (H2) evolution. However, to date, little work has been done regarding the decoration approaches to hybridize TMPs on the g-C3N4 surface with homogeneous dispersion and intimate interfacial contact. Herein, we present a facile and convenient route to in situ incorporate g-C3N4 nanosheets (NSs) and Ni2P nanocrystal (NC) co-catalysts via a one-step co-heating solution approach. The Ni2P/g-C3N4 (in situ) hybrid photocatalyst achieved a far superior H2 production rate (2849.5 μmol g−1 h−1) and durability (no decrease after 4 cycles of reaction within 20 h) compared to the Ni2P/g-C3N4 (self-assembly) sample. The apparent quantum yield (AQY) of 18.8% at 420 nm was also much higher than that of other TMP co-catalyst loaded g-C3N4 hybrid photocatalysts. A possible Ni(δ+)–N(δ−) chemical coupling in the Ni2P/g-C3N4 (in situ) hybrid composite was proposed and corroborated by X-ray photoelectron spectroscopy (XPS) and X-ray absorption spectroscopy. The unique Ni(δ+)–N(δ−) chemical bonding states between g-C3N4 and Ni2P significantly accelerate the photo-generated charge-carrier separation and extraction from g-C3N4, as well as maintaining the H2 production durability. We believe that the hybridization route presented in this work will be extended to construct other TMP integrated photocatalysts toward efficient and stable solar water splitting.


 Please wait while we load your content...
Please wait while we load your content...