Exploring the properties and potential biomedical applications of NSAID-capped peptide hydrogels
Abstract
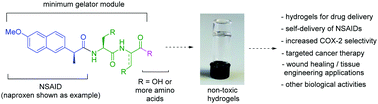
The development of strategies to minimise the adverse side-effects of non-steroidal anti-inflammatory drugs (NSAIDs) remains a challenge for medicinal chemists. One such strategy is the development of NSAID–peptide prodrug conjugates and this conjugation to a peptide often confers the additional property of hydrogelation. This review summarises the work published by our research group, alongside other research groups, on supramolecular hydrogels consisting of short peptides conjugated to NSAIDs. Generally, supramolecular low molecular weight hydrogels (LMWHs) are composed of amphiteric molecules, usually consisting of short peptides attached to an aromatic capping group. When the aromatic capping group is switched for an NSAID to afford hybrid gelators, some conjugates exhibit retained or improved anti-inflammatory properties of the parent drug, and sometimes new and unexpected biological activities are observed. Conjugation to peptides often provides selective COX-2 inhibition over COX-1 inhibtion, which is key to retaining the anti-inflammatory benefits of NSAIDs whilst minimising gastric side-effects. Naproxen is the most commonly employed NSAID capping group, partly due to its similarity in structure to commonly employed naphthalene capping groups. Biomimetic approaches, where canonical amino acids are switched for non-natural amino acids such as D-amino acids or dehydroamino acids, are often employed, to tune the stability. The future direction for this area of research is discussed.
- This article is part of the themed collection: Peptide Soft Materials


 Please wait while we load your content...
Please wait while we load your content...