Effects of fluoro substitutions and electrostatic interactions on the self-assembled structures and hydrogelation of tripeptides: tuning the mechanical properties of co-assembled hydrogels†
Abstract
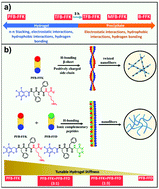
A series of FFK tripeptides capped with phenylacetic acid of various fluoro-substitutions at the N-terminus has been synthesized and examined for self-assembly under aqueous conditions. The material properties of the FFK tripeptides dramatically changed from precipitate to hydrogel phase upon increasing the number of fluorine atoms. Peptides linked with benzyl (B-FFK) or monofluoro-benzyl (MFB-FFK) groups rapidly form solid precipitates under physiological pH conditions. The trifluoro-decorated compound (TFB-FFK) self-assembled into a metastable hydrogel which slowly transformed into a solid precipitate upon standing. A stable hydrogel formation was noticed in the case of the pentafluorobenzyl-diphenylalanyllysine (PFB-FFK) compound. TEM analysis indicates that the PFB-FFK peptide assembled into twisted nanofibril structures, which are predominantly stabilized by strong quadrupole π-stacking interactions and electrostatic interactions of amino acid side chains. Furthermore, the combination of PFB-FFK and PFB-FFD peptides was also investigated for hydrogelation and the self-assembly of such systems resulted in the formation of untwisted 1D nanofibril structures. Supramolecular coassembled hydrogels of variable stiffness have also been achieved by modulating the concentration of the peptide components, which was evident from the rheological analysis. Such low molecular weight (LMW) peptide materials with tuneable mechanical properties might be a potential material for a wide range of applications in nanotechnology and biotechnology.
- This article is part of the themed collection: Peptide Soft Materials


 Please wait while we load your content...
Please wait while we load your content...