The self-diffusion of polymethylsilsesquioxane (PMSSO) dendrimers in diluted solutions and melts†
Abstract
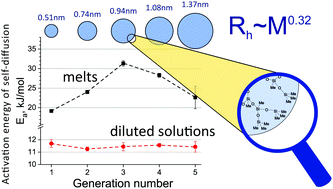
Recently developed non-functional derivatives of polymethylsilsesquioxane (PMSSO) dendrimers of the first to fifth generation were characterized by 1H, 13C and 29Si NMR spectroscopy. The self-diffusion and NMR relaxation of PMSSO dendrimers in dilute solutions of toluene and melts were investigated in a wide temperature range (−50–80 °C). The hydrodynamic radii of dendrimers were determined from the self-diffusion coefficients measured in diluted solutions according to the Stokes–Einstein equation. The hydrodynamic radius of PMSSO dendrimers as a function of molecular mass follows a power law with the scaling exponent of 0.32 ± 0.02 in the investigated temperature range. The temperature dependences of the self-diffusion coefficients of dendrimers were described by the Arrhenius-type equation. The activation energies of self-diffusion of dendrimers in diluted toluene solutions are identical for different generations while the dependence of activation energy for dendrimers in melts shows a maximum for the third generation (G3) dendrimer. Taking into account the absence of specific interactions in PMSSO dendrimer melts the observed behavior was ascribed to the manifestation of interpenetration of dendrimer molecules. For low generations (G1 and G2) the short length of the branches does not considerably affect the translational diffusion while for higher generations (G4 and G5) the densification of the structure prevents significant interpenetration.


 Please wait while we load your content...
Please wait while we load your content...