Packing and dynamics of a protein solution approaching the jammed state†
Abstract
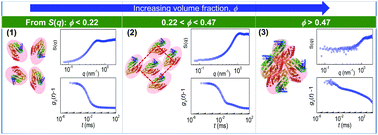
The packing of proteins and their collective behavior in crowded media is crucial for the understanding of biological processes. Here we study the structural and dynamical evolution of solutions of the globular protein bovine serum albumin with increasing concentration via drying using small angle X-ray scattering and dynamic light scattering. We probe an evolving correlation peak on the scattering profile, corresponding to the inter-protein distance, ξ, which decreases following a power law of the protein volume fraction, ϕ. The rate of decrease in ξ becomes faster above a protein concentration of ∼200 mg ml−1 (ϕ = 0.15). The power law exponent changes from 0.33, which is typical of colloidal or protein solutions, to 0.41. During the entire drying process, we observe the development and the growth of two-step relaxation dynamics with increasing ϕ as revealed by dynamic light scattering. We find three different regimes of the dependence of ξ as a function of ϕ. In the dilute regime (ϕ < 0.22), protein molecules are far apart from each other compared to their size. In this case, the dynamics mainly corresponds to Brownian motion. At an intermediate concentration (0.22 < ϕ < 0.47), inter-protein distances become comparable to the size of protein molecules, leading to a preferential orientation of the ellipsoidal protein molecules along with a possible deformation. In this regime, the dynamics shows two distinct relaxation times. At a very high concentration (ϕ > 0.47), the system reaches a jammed state. Subsequently, the secondary relaxation time in this state becomes extremely slow. In this state, the protein molecules have approximately one hydration layer. This study contributes to the understanding of protein molecular packing in crowded environments and the phenomenon of density-driven jamming for soft matter systems.


 Please wait while we load your content...
Please wait while we load your content...