Membrane determinants for the passive translocation of analytes through droplet interface bilayers†
Abstract
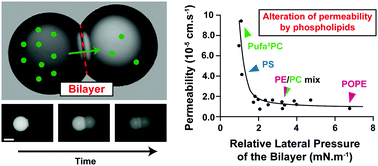
Understanding how small molecules cross cell membranes is crucial to pharmaceutics. Several methods have been developed to evaluate such a process, but they need improvement since many false-positive candidates are often selected. Robust tools enabling rapid and reproducible screening can increase confidence on hits, and artificial membranes based on droplet interface bilayers (DIBs) offer this possibility. DIBs consist in the adhesion of two phospholipid-covered water-in-oil droplets which reproduce a bilayer. By having donor and acceptor droplets, the permeability of an analyte can be studied. However, the relevance of this system relies on the comprehension of how well the physical chemistry of the produced bilayer recapitulates the behavior of cell membranes. This information is missing, and we address it here. Taking small fluorophores as model analytes, we studied their permeation through DIBs made of a wide range of phospholipids. We found that both the phospholipid acyl chain and polar head affect permeability. Overall, these parameters impact the phospholipid shape and thereupon the membrane lateral pressure, which is a major factor modulating with permeability in our system. These results depend on the nature of the chosen oil. We thereupon identified relevant physical chemistry conditions that best mimic the compactness and subsequent permeability of biological membranes.


 Please wait while we load your content...
Please wait while we load your content...