Controlling protein adsorption modes electrostatically
Abstract
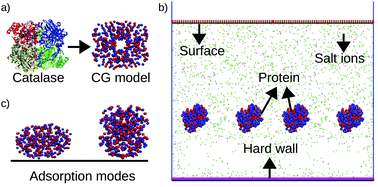
Protein adsorption on surfaces is ubiquitous in biology and in biotechnology. There are various forces required for controlling protein adsorption. Here, we introduce an explicit ion coarse-grained molecular dynamics simulation approach for studying the effects of electrostatics on protein adsorption, and 2D protein assembly on charged surfaces. Our model accounts for the spatial distribution of protein charges. We use catalase as our model protein. We find that the preferential adsorption mode of proteins at low protein concentration on a charged surface is “standing up”. When the protein concentration in a solution increases to reach a critical density on the surface, the adsorption mode switches from “standing up” to a mixed state “flat on” and “standing up”, which increases the lateral correlations among the adsorbed proteins. As such, the changes in the adsorption mode arise from the protein adsorption that cancel the surface charge and the protein–protein repulsion. This correlated surface structure melts as the salt concentration increases because the charged surface is cancelled by the salt ions and the proteins de-adsorb. For the case of strongly charged surfaces the “standing up” conformation remains more favorable even at high protein adsorption at low salt concentrations since in that conformation the surface charge is cancelled more effectively, generating an even more laterally correlated structure. We elucidate the effects of parameters such as surface charge density, salt concentration, and protein charges on the different adsorption modes and the structure and organization of proteins on the charged surfaces. This study provides a guide for controlling protein assembly on surfaces.


 Please wait while we load your content...
Please wait while we load your content...
