Leidenfrost phenomenon during quenching in aqueous solutions: effect of evaporation-induced concentration gradients
Abstract
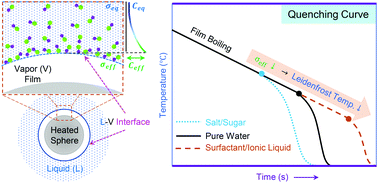
The minimum temperature limit for a sustained vapor film on a hot surface defines the well-known Leidenfrost temperature (LFT). LFT for pure fluids is typically a strong function of the surface tension. However, the effect of surface tension on LFT of aqueous additive solutions is confusing with many complicated trends. For example, despite an insignificant increase of ≈1 mN m−1 in surface tension, a substantial increase in LFT of ≈50 °C with aqueous salt and sugar solutions has been reported in comparison to pure water. Conversely, no appreciable change in LFT (within ±2 °C) is observed despite a substantial drop of up to ≈30 mN m−1 in surface tension upon varying the concentration of surfactant additives in aqueous solutions. Here, we perform simultaneous thermal, visual, and acoustic characterization of pool quenching experiments with aqueous solutions of salt, sugar, surfactant, and ionic liquids. We model the evaporation-induced increase in the concentration of the non-volatile additives at the liquid–vapor interface using Fick's second law of diffusion. We show that the localized concentration buildup of additives at the liquid–vapor interface dramatically alters the surface tension values in comparison to the typical equilibrium values estimated otherwise. We use these modified surface tension values to correlate the diverse set of experimental LFT data reported in our work and in the literature using a unified framework. We believe that these clarifications regarding the Leidenfrost mechanism will encourage the use of additives in various applications, specifically those where surface modification strategies may not be practically feasible.


 Please wait while we load your content...
Please wait while we load your content...