3D aggregation of cells in packed microgel media†
Abstract
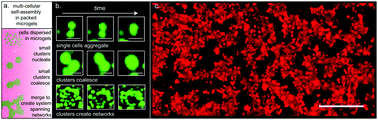
In both natural and applied contexts, investigating cell self-assembly and aggregation within controlled 3D environments leads to improved understanding of how structured cell assemblies emerge, what determines their shapes and sizes, and whether their structural features are stable. However, the inherent limits of using solid scaffolding or liquid spheroid culture for this purpose restrict experimental freedom in studies of cell self-assembly. Here we investigate multi-cellular self-assembly using a 3D culture medium made from packed microgels as a bridge between the extremes of solid scaffolds and liquid culture. We find that cells dispersed at different volume fractions in this microgel-based 3D culture media aggregate into clusters of different sizes and shapes, forming large system-spanning networks at the highest cell densities. We find that the transitions between different states of assembly can be controlled by the level of cell–cell cohesion and by the yield stress of the packed microgel environment. Measurements of aggregate fractal dimension show that those with increased cell–cell cohesion are less sphere-like and more irregularly shaped, indicating that cell stickiness inhibits rearrangements in aggregates, in analogy to the assembly of colloids with strong cohesive bonds. Thus, the effective surface tension often expected to emerge from increased cell cohesion is suppressed in this type of cell self-assembly.
- This article is part of the themed collection: Liquid Composites


 Please wait while we load your content...
Please wait while we load your content...