Amphiphilic drug–peptide–polymer conjugates based on poly(ethylene glycol) and hyperbranched polyglycerol for epidermal growth factor receptor targeting: the effect of conjugate aggregation on in vitro activity†
Abstract
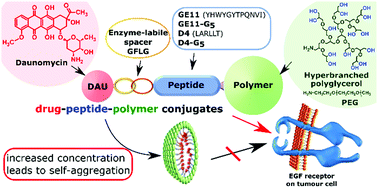
Numerous peptide–drug conjugates have been developed over the years to enhance the specificity and selectivity of chemotherapeutic agents for tumour cells. In our present work, epidermal growth factor receptor targeting drug–peptide conjugates were prepared using GE11 and D4 peptides. To ensure the drug release, the cathepsin B labile GFLG spacer was incorporated between the targeting peptide and the drug molecule (daunomycin), which significantly increased the hydrophobicity and thereby decreased the water solubility of the conjugates. To overcome the solubility problem, drug–peptide–polymer conjugates with systematic structural variations were prepared, by linking poly(ethylene glycol) (PEG) or a well-defined amino-monofunctional hyperbranched polyglycerol (HbPG) directly or via a pentaglycine spacer to the targeting peptides. All the drug–peptide–polymer conjugates were water-soluble as confirmed by turbidimetric measurements. The results of the in vitro cell viability and cellular uptake measurements on HT-29 human colon adenocarcinoma cells proved that the HbPG and the PEG highly influenced the biological activity. The conjugation of the hydrophilic polymer resulted in the amphiphilic character of the conjugates, which led to self-aggregation and nanoparticle formation that decreased the cellular uptake above a specific aggregation concentration. On the other hand, the hydrodynamic volume and the different polymer chain topology of the linear PEG and the compact hyperbranched HbPG also played an important role in the biological activity. Therefore, in similar systems, the investigation of the colloidal properties is inevitable for the better understanding of the biological activity, which can reveal the structure–activity relationship of amphiphilic drug–peptide–polymer conjugates for efficient tumour targeting.


 Please wait while we load your content...
Please wait while we load your content...