Electrophoretic origin of long-range repulsion of colloids near water/Nafion interfaces†
Abstract
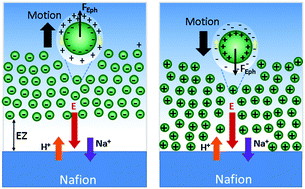
One of the most striking properties of Nafion is the formation of a long-range solute exclusion zone (EZ) in contact with water. The mechanism of formation of this EZ has been the subject of a controversial and long-standing debate. Previous studies by Schurr et al. and Florea et al. root the explanation of this phenomenon in the ion-exchange properties of Nafion, which generates ion diffusion and ion gradients that drive the repulsion of solutes by diffusiophoresis. Here we have evaluated separately the electrophoretic and chemiphoretic contributions to multi-ionic diffusiophoresis using differently charged colloidal tracers as solutes to identify better their contribution in the EZ formation. Our experimental results, which are also supported by numerical simulations, show that the electric field, built up due to the unequal diffusion coefficients of the exchanged ions, is the dominant parameter behind such interfacial phenomenon in the presence of alkali metal chlorides. The EZ formation depends on the interplay of the electric field with the zeta potential of the solute and can be additionally modulated by changing ion diffusion coefficients or adding salts. As a consequence, we show that not all solutes can be expelled from the Nafion interface and hence the EZ is not always formed. This study thus provides a more detailed description of the origin and dynamics of this phenomenon and opens the door to the rational use of this active interface for many potential applications.


 Please wait while we load your content...
Please wait while we load your content...