Compaction and self-association of megabase-sized chromatin are induced by anionic protein crowding†
Abstract
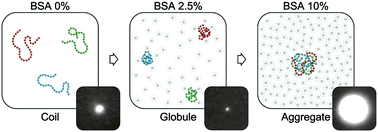
Highly compacted chromatin, a complex of DNA with cationic histone proteins, is found in the nucleus of eukaryotic cells in an environment with a high concentration of macromolecular species, many of which possess a negative charge. In the majority of previous studies, however, these crowding conditions were experimentally modelled using neutral synthetic macromolecules such as polyethylene glycol (PEG). Despite the importance of the crowding agent charge in the condensation process of chromatin, to the best of our knowledge, the behavior of chromatin under conditions of anionic protein crowding has not been studied. Here, compaction of nearly megabase-long chromatin in the presence of the anionic globular protein BSA was investigated by single-molecule fluorescent microscopy (FM). We demonstrate different effects of anionic macromolecular crowders (MMCs) on DNA and chromatin, compared to neutral MMCs. While DNA molecules undergo gradual compaction into a globular form in the presence of ca. 20% w/v of BSA, chromatin fibres complete coil to globule transition at a much lower concentration of BSA (ca. 5% w/v). Furthermore, at higher concentrations of BSA in solution (>5% w/v), chromatin fibres self-associate and form large spherical or fibrillar supramolecular microstructures characterized by a high colloidal stability and dynamic intermolecular fluctuations. Formation of such self-organized colloids from chromatin is universal and characteristic of chromatin fibres of various lengths. Our results highlight the hitherto underappreciated effect of anionic MMC environment on chromatin higher-order structures that may play an important role in self-organization of chromatin in vivo.


 Please wait while we load your content...
Please wait while we load your content...