Insight into the formation and permeability of ionic liquid unilamellar vesicles by molecular dynamics simulation†
Abstract
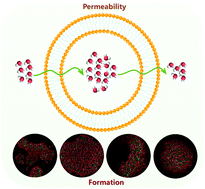
Unilamellar vesicles in solution could open up new horizons for reaction and material delivery, but the formation mechanism especially for the permeability of the small molecule through the vesicle membrane is still unknown. In this study, the formation and permeability of the unilamellar vesicles formed by the ionic liquid 1-dodecyl-3-methylimidazolium salicylate ([C12mim][Sal]) have been investigated by molecular dynamics simulation. Starting from a random distribution of ionic liquids, the entire process of vesicle formation could be observed on a nanosecond time scale, during which planar and cup-like structures are formed at the intermediate stage. Energy analysis reveals that the electrostatic interactions between cations and anions play a dominant role in forming and stabilizing the vesicle. Radial density distribution functions indicate that the final stable vesicle is a spherical bilayer structure. Besides, it was found that the structure of vesicles is maintained with the increase of temperature, while the water molecules in the vesicles could be completely exchanged quickly. These results suggest that vesicles may be beneficial for the enrichment or release of molecules.


 Please wait while we load your content...
Please wait while we load your content...