Thermodynamic control over energy dissipation modes in dual-network hydrogels based on metal–ligand coordination
Abstract
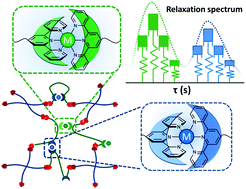
Modern polymeric hydrogels use reversible bonds to mimic biological functionalities. However, true biological materials benefit from several supramolecular elements and deliver multiple functions at the same time. To approach similar creation and control of multiple different functional elements in a synthetic soft material, we develop a model dual-network hydrogel in which multiple energy dissipating modes are formed by metal–ligand coordination and regulated by their association thermodynamics. This idea is realized by using linear and tetra-arm poly(ethylene glycol) (PEG) precursors with complementary reactive end groups. The former also carries terpyridine ligands on both ends, which form metallo-supramolecular bonds upon addition of metal ions. Multiple relaxation modes are provided by a combination of different metal ions. The timescale and amplitude of energy dissipating elements are characterized by oscillatory shear deformation. These studies suggest that the composition of metal ions controls the contribution of the corresponding relaxation modes in a linear fashion. A molecular-level confirmation is provided by following the UV-vis absorbance of the linear precursor in combination with mixtures of metal ions, accompanied by a theoretical study on the kinetics of the reversible association process. These results show that the linearity of the aforementioned dependence holds for such systems in which the utilized combination of metal ions and ligands exclusively form stable bis-complexes. By contrast, in many other cases, especially when the ions may compete to form mono-, bis-, or tris-complexes with the ligand, deviation from linearity is expected.
- This article is part of the themed collection: Soft Matter Most Popular 2020


 Please wait while we load your content...
Please wait while we load your content...