Hydrogen via reforming aqueous ammonia and biomethane co-products of wastewater treatment: environmental and economic sustainability
Abstract
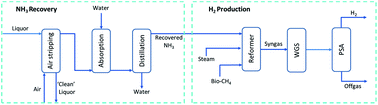
Green H2 is increasingly viewed as a key energy carrier for the fight against climate change. Wastewater treatment plants (WWTPs) have the unique potential to be centres of renewable H2 generation with the growing availability of two attractive feedstocks: biomethane and ammonia. An innovative and novel method of ammonia recovery from digestate liquor followed by a state-of-the-art H2 production process named NWaste2H2 is demonstrated for a case-study WWTP. The recovered ammonia is used alongside biomethane for H2 production and its diversion from conventional biological treatment has two other crucial benefits, with reductions in both associated electricity demand and emissions of nitrous oxide, an extremely potent greenhouse gas. Process modelling, supported by extensive experiments in a packed-bed reactor at bench-scale, demonstrate the prized capability of simultaneously performing steam methane reforming and ammonia decomposition to generate a H2-rich syngas with yields close to equilibrium values. Greenhouse gas emission abatement from the replacement of diesel buses and reduced N2O emissions from biological treatment could save up to 17.2 kg CO2 equivalent (CO2e) per year for each person served by the WWTP. An in-depth economic study illustrates the ability to achieve a positive net present value with a 10% discount factor as early as 5.8 years when the H2 is prepared and sold to power fuel cell electric buses.


 Please wait while we load your content...
Please wait while we load your content...