Hydrogen-efficient non-oxidative transformation of methanol into dimethoxymethane over a tailored bifunctional Cu catalyst†
Abstract
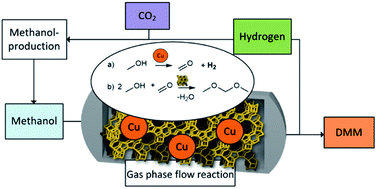
Dimethoxymethane (DMM), a promising synthetic fuel enabling clean combustion, is usually produced by condensation of methanol and formaldehyde, where the latter stems from methanol oxidation. Here, we report the hydrogen efficient non-oxidative DMM synthesis over a bifunctional Cu/zeolite catalyst in a continuous gas-phase fixed bed reactor. Methanol dehydrogenation to formaldehyde (FA) is coupled with FA condensation with methanol to yield DMM, hydrogen and water. Thermodynamic analysis confirms the general feasibility of this route and also manifests the vital importance of catalyst selectivity. Therein, close proximity of the catalyst's metallic Cu species and acidic sites is crucial. Noticeably, DMM selectivity of the catalyst only evolves within the first 13 hours of operation rising from 5.8 to 77.2%. A maximum DMM selectivity of 89.2 or 80.3% could be reached for 0.4 and 0.7 wt% Cu on Hβ(836) zeolite with 1.9 or 3.6% methanol conversion, respectively. Comprehensive characterizations emphasize adaptation of Cu species and Hβ zeolite under reaction conditions resulting in the decisive weakened dehydrogenation and condensation ability for high DMM selectivity. Process simulations confirm superior exergy efficiency compared to state-of-the-art technologies for DMM production already with the herein developed catalyst and highlights the high potential of further innovations for technical implementation.


 Please wait while we load your content...
Please wait while we load your content...