Understanding the effect of H2O on CO2 adsorption capture: mechanism explanation, quantitative approach and application†
Abstract
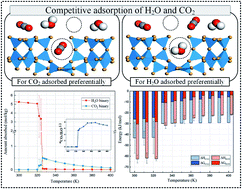
The presence of water vapor in realistic flue gas should be emphasized in the development of adsorbents for post-combustion CO2 capture. However, the co-adsorption mechanisms of H2O and CO2 molecules on adsorbents are still unclear. In this paper, the adsorption performance of CO2 under wet flue gas was investigated through molecular simulation. The mechanism of H2O being preferentially adsorbed at low temperature and CO2 being preferentially adsorbed at high temperature has been clarified. In order to further provide a quantitative criterion, a calculation method was introduced. The Gibbs free adsorption energy was applied to explain the competitive adsorption behavior of CO2 and H2O. The adsorption entropy and the adsorption enthalpy were derived according to the statistical thermodynamics. Molecular dynamics simulation is the main research method in this paper. A proper force field for H2O was identified among SPC/E, TIP4P, and TIP5P models. The adsorption isotherms of CO2, H2O, and CO2/H2O were obtained by Grand Canonical Monte Carlo (GCMC) simulations from 300 K to 400 K. A case study for CO2/H2O mixture adsorption on zeolite MFI is presented to demonstrate the feasibility. The obtained equilibrium adsorption isotherms from the case study showed that the presence of a small amount of H2O in the gas mixture had a significant impact on the adsorption loads of CO2. A detailed thermodynamics analysis demonstrated that the adsorption entropies and enthalpies were temperature dependent and they would switch the relative order of Gibbs free adsorption energy between adsorbed CO2 and H2O. Moreover, the CO2 working capacity and energy consumption of regeneration in the process of temperature swing adsorption (TSA) were presented.
- This article is part of the themed collection: Sustainable Energy & Fuels Cover Art


 Please wait while we load your content...
Please wait while we load your content...