Photoelectrochemical conversion of CO2 into HCOOH using a polymeric carbon nitride photoanode and Cu cathode†
Abstract
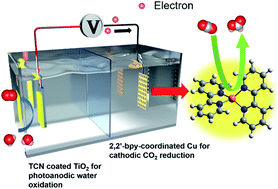
A photoelectrocatalysis (PEC) system is developed for HCOOH production by the reduction of CO2 that is achieved by light illumination without any sacrificial agent. The photoanode is based on a polymeric carbon nitride semiconductor for water oxidation releasing electrons and protons to reduce CO2 on the cathode of Cu mesh coordinated with 2,2′-bipyridine. The Cu and 2,2′-bpy coordination promotes the adsorption of CO2 and the directional transport of electrons, thereby increasing the selectivity of HCOOH. In the two-electrode system, the photoelectrodes are wired with an external power supply to achieve a PEC system for sustainable production of HCOOH from atmospheric CO2 under ambient conditions. In this system, the performance of light-to-HCOOH can be preserved for least 16 hours, demonstrating the high stability of the conjugated polymers against photooxidation corrosion. This development offers opportunities for scaling up the application of fixation of CO2 using all sustainable materials.


 Please wait while we load your content...
Please wait while we load your content...