Well-ordered Cs–Ru/@SBA-15 nanocomposite materials for low pressure ammonia synthesis†
Abstract
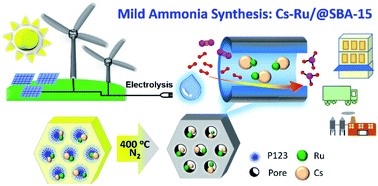
A facile route for preparing large-pore SBA-15-supported and Cs-promoted Ru nanocomposite materials (denoted as Cs–Ru/@SBA-15) was developed for catalysing the synthesis of ammonia under low pressure conditions. Aqueous solutions of ruthenium(III) nitrosyl nitrate and caesium carbonate were impregnated on the as-prepared SBA-15 mesoporous silica in a step-wise manner. The P123 template not only chelated with the Ru and Cs species but also stabilised the mesoporous silica framework, prior to carbonisation at 400 °C in N2. As a result, the Cs–Ru/@SBA-15 catalysts prepared with Ru loadings of 2–6 wt% and a fixed Cs/Ru molar ratio of 2.5 possessed high surface area, porosities, and ordered pore structures. The XRD patterns, CO chemisorption data, and STEM images revealed that the nanostructured Ru particles (<5 nm) and amorphous Cs species were stably embedded within the channelling pores of the prepared Cs–Ru/@SBA-15 catalysts when the Ru loadings were below 8 wt%. The studies of temperature-programmed reduction and surface reaction demonstrated that there was a strong interaction between the Ru and Cs species, which contributed to high ammonia synthesis activity (>1.5 mmol NH3 gcat−1 h−1) of the Cs–Ru/@SBA-15 catalysts at atmospheric pressure. Conversely, the catalysts impregnated with only Ru or Cs species had no effect on the ammonia synthesis. The solid 13Cs–4Ru/@SBA-15 catalyst with a Ru loading of 3.2 wt% and Cs/Ru molar ratio of 3.1 exhibited the highest activity (5.3 mmol NH3 gcat−1 h−1) at 460 °C and a H2/N2 molar ratio of 1 at atmospheric pressure. The high activity was maintained by varying the space velocity, H2/N2 molar ratio, and reaction temperature under low-pressure conditions, which facilitated green ammonia synthesis using low-carbon hydrogen derived from renewable energy-based water hydrolysis.


 Please wait while we load your content...
Please wait while we load your content...