A comparative study of undoped, boron-doped, and boron/fluorine dual-doped carbon nanoparticles obtained via solution plasma as catalysts for the oxygen reduction reaction†
Abstract
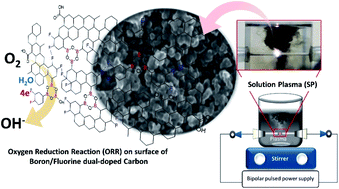
Boron/fluorine dual-doped carbons, which have never been reported for their oxygen reduction reaction (ORR) activity, were successfully synthesized through a one-step solution plasma (SP) process using mixtures of toluene, as a carbon source, and heterocyclic compounds containing boron and fluorine atoms, i.e., 2,4,6-tris(4-fluorophenyl)boroxin, 2,4,6-tris(3,4-difluorophenyl)boroxin and 2,4,6-tris(3,4,5-trifluorophenyl) boroxin. Therefore, in this study, the obtained undoped, boron-doped, and boron/fluorine dual-doped carbons, synthesized using SP, were investigated and compared to find their catalytic activity toward the ORR. The FE-SEM observation showed that the obtained carbons had a diameter in the nanoscale range. XRD and Raman measurements revealed that the synthesis via SP led to the generation of turbostratic carbons with structural defect sites. The carbon atoms were found to mainly interact with fluorine atoms through semi-ionic C–F bonding. All SP-synthesized carbon nanoparticles showed acceptable ORR activity in alkaline solution. However, boron/fluorine dual-doped carbon nanoparticles also drove the ORR through a two-electron pathway with low generation of the unstable peroxide, HO2−, compared to undoped and boron-doped carbon nanoparticles.


 Please wait while we load your content...
Please wait while we load your content...
