Ru–Fe nanoalloys supported on N-doped carbon as efficient catalysts for hydrogen generation from ammonia borane
Abstract
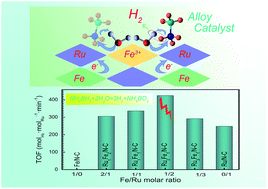
Hydrogen is a potential clean energy carrier for renewable energy sources. The development of high-efficiency and economical catalysts for hydrogen generation is of great importance. In this work, highly dispersed RuFe alloys were prepared and anchored on the surface of microsphere-type N-doped carbon through a facile impregnation-co-reduction process. The spherical N-doped carbon derived from a resorcinol–formaldehyde resin effectively promoted the surface dispersion of the RuFe alloys and the distribution of catalytic active sites on the support surface. According to the density functional theory simulation, the significant positive synergistic effect originated from the coexistence of Ru and Fe in the alloy and effectively reduced the reaction energy barrier. The alloys with various Ru/Fe ratios were more catalytically active for ammonia borane hydrolysis than their supported single Ru or Fe counterparts. This alloy strategy also effectively improved the utilization ratio and stability of Ru. With the optimized Ru2Fe1/N–C catalyst, an unprecedented hydrogen generation specific rate of 9.4 × 104 mL min−1 gRu−1 was achieved at 298 K. The catalytic activity tended to be stable after five consecutive uses. This work provides a beneficial reference for the design of high-efficiency catalysts for hydrogen generation.


 Please wait while we load your content...
Please wait while we load your content...