Trimetallic CoFeCr hydroxide electrocatalysts synthesized at a low temperature for accelerating water oxidation via tuning the electronic structure of active sites†
Abstract
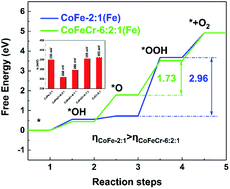
Efficient methods are urgently required for developing electrocatalysts with superior activities and practical applications. In this work, a novel pure ternary CoFeCr hydroxide was synthesized on carbon paper using a low-temperature (60 °C) hydrothermal approach. The OER performances of the CoFeCr hydroxides with different atomic ratios were systematically investigated. Intriguingly, the optimized trimetallic CoFeCr-6:2:1 electrocatalyst exhibited significantly improved OER performance with a small overpotential of 260 mV at 10 mA cm−2 and a low Tafel slope of 40.1 mV dec−1 under alkaline conditions. Combined experimental and DFT+U computational studies revealed that Fe was the active site and the excellent OER activity could be attributed to the electronic structure regulation effect via the introduction of Cr, which on the one hand improved the d-band center position of the Fe sites and on the other hand decreased the band gap of the sample. This regulation effect optimized the adsorption of oxygen species and efficiently accelerated the oxygen evolution kinetics and charge transfer kinetics, thus enhancing the OER performance of CoFeCr-6:2:1. Our results provide both experimental and theoretical basis for the further exploration of efficient hydroxide electrocatalysts for energy-related applications.


 Please wait while we load your content...
Please wait while we load your content...