Engineering the surface wettability of a ceramic carbon electrode for improved hydrogen evolution performance of a molybdenum sulfide electrocatalyst†
Abstract
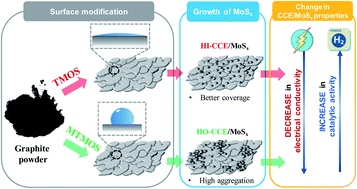
In the present work, we explored an economically feasible and environmentally friendly attempt to achieve tunable wettability of ceramic carbon substrates applicable in water splitting devices. It was found that by rationally controlling the surface functional groups, ceramic carbon substrates with different surface hydrophilicities, electroactive surface areas, and electrochemical behaviors could be achieved. Particularly, a hydrophilic ceramic carbon electrode (CCE) was obtained by using tetramethoxysilane as the modifier, which allowed uniform anchoring of an amorphous molybdenum sulfide (MoSx) hydrogen evolution electrocatalyst on its surface through a simple chemical vapor deposition method. The optimized CCE/MoSx exhibited promising hydrogen evolution reaction activity and good stability, with a small onset overpotential (∼250 mV) and a low Tafel slope (58 mV dec−1). The CCE/MoSx catalyst also showed reasonable stability under acidic conditions. Such promising electrochemical performance has been attributed to the strong chemical and electronic coupling between the conducting CCE and MoSx. This work paves a new way for the rational design and fabrication of an efficient composite electrode by selecting ceramic carbon as a durable high-conductivity substrate and the collaborative optimization of the catalyst and the substrate to achieve higher hydrogen evolution performance.


 Please wait while we load your content...
Please wait while we load your content...