From non-innocent to guilty: on the role of redox-active ligands in the electro-assisted reduction of CO2 mediated by a cobalt(ii)-polypyridyl complex†
Abstract
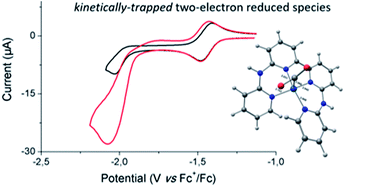
The electrochemical behavior of [Co(bapbpy)Cl]+ [1-Cl]+, a pentacoordinated polypyridyl cobalt(II) complex containing a redox-active tetradentate ligand (bapbpy: 6,6′-bis-(2-aminopyridyl)-2,2′-bipyridine) has been investigated in DMF. Cyclic voltammograms (CV), recorded in the presence of increasing amounts of chloride anions, highlighted the existence of an equilibrium with the neutral hexacoordinated complex. Under a CO2 atmosphere, CVs of [Co(bapbpy)Cl]+ exhibit significant current enhancement assigned to CO2 catalytic reduction. Controlled-potential electrolysis experiments confirmed formation of CO and HCOOH as the only identifiable products. The addition of water or chloride ions was shown to affect the distribution of the products obtained, as well as the faradaic efficiency associated with their electrocatalytic generation. A combination of electrochemical techniques, chemical reductions, spectroscopic measurements (UV-vis and IR) and quantum chemical calculations suggests that the ability of the bapbpy ligand to be reduced at moderately negative potentials drastically limits the catalytic performances of [1-Cl]+, by stabilizing the formation of a catalytically-competent CO2-adduct that only slowly reacts with oxide acceptors to evolve towards the desired reduction products.


 Please wait while we load your content...
Please wait while we load your content...