Industrial feasibility of anodic hydrogen peroxide production through photoelectrochemical water splitting: a techno-economic analysis†
Abstract
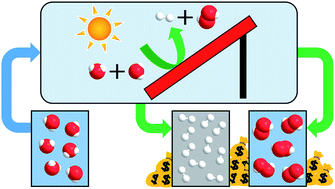
Photoelectrochemical (PEC) water splitting is a promising approach to drive green, carbon-free production of hydrogen (H2). In ‘classic’ water splitting, oxygen (O2) is formed at the anode as a by-product. It has been suggested that substitution of anodic O2 production with hydrogen peroxide (H2O2) could increase the financial attractiveness of PEC water splitting. Here, we present a techno-economic analysis of a photoelectrochemical H2/H2O2 process. Specifically, we model photoelectrochemical farms with industrially relevant production capacities. Two scenarios are considered: (i) a theoretical scenario with an optimal solar-to-hydrogen (STH) efficiency of 27.55% and (ii) a literature-based state-of-the-art scenario with an STH efficiency of 10.1%. When applying an averaged market value of $0.85 kg−1 for H2O2, the analysis reveals a negative levelized cost of hydrogen (LCH) for scenario (i), i.e. $6.45 kg−1, and for scenario (ii) an LCH of $6.19 kg−1. Our results imply that these values are superior to the LCH of ‘classic’ PEC water splitting (ca. $10 kg−1), while the negative value for scenario (i) even outcompetes the LCH of steam methane reforming ($1.4 kg−1). We predict that significant reduction in the LCH can be realized within the PEC community when future research is aimed at enhancing the stability of the photoanode and optimizing the STH efficiency for anodic H2O2 formation. This manuscript clearly demonstrates the financial benefits of value-added product formation, such as hydrogen peroxide, over O2 formation. In a broader context, our analysis verifies that further research on valuable commodity chemicals at the anode in water splitting and CO2 reduction should be stimulated in the future to facilitate implementation of emerging, cost-intensive technologies.


 Please wait while we load your content...
Please wait while we load your content...