Dynamics of carbon formation during the catalytic hydrodeoxygenation of raw bio-oil†
Abstract
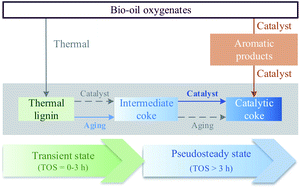
The formation, growth and transformation of the carbon residue (coke) deposited on the catalyst during the raw bio-oil hydrodeoxygenation have been studied. These deposits have a great impact on the overall process performance, and they have been formed in accelerated deactivation conditions (450 °C, 65 bar, space time of 0.09 gcat h gbio-oil−1) using a continuous fixed bed reactor and a FeMoP/HZSM-5 catalyst. Coke deposition causes partial deactivation of the catalyst, which reaches a pseudosteady state of constant activity and also contant yields of interesting chemicals. The evolution of the coke in the transient state has been studied through temperature-programmed oxidation, Raman spectroscopy and elemental analysis. We have identified three different types of coke, whose composition evolves with time on stream towards condensed and stable structures. The assessment of the evolution of the reaction medium composition and the application of the principal component analysis (PCA) methodology have evidenced that the dynamics of coke have three stages: (1) it is controlled by the thermally-induced deposition of thermal lignin; (2) followed by the interconversion into intermediate coke through aging reactions; and (3) it ends up in a pseudosteady state dominated by the formation of catalytic coke species originating from both deoxygenated and carbonized intermediate coke as well as the condensation of aromatics in the reaction medium.
- This article is part of the themed collection: Sustainable Energy & Fuels Cover Art


 Please wait while we load your content...
Please wait while we load your content...