Oxidized indium with transformable dimensions for CO2 electroreduction toward formate aided by oxygen vacancies†
Abstract
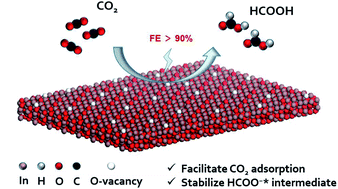
Electroreduction of CO2 into value-added products is a promising technique in which the structure of the catalyst plays a crucial role. Herein, the structural evolution of low-dimensional oxidized indium catalysts and the impact of surface oxygen vacancies on the catalytic efficiency were investigated. We first utilize in situ environmental transmission electron microscopy (ETEM) to monitor the transformation of two-dimensional InOOH nanosheets into oxygen-vacancy-rich (Vo-rich) InOOH nanosheets and then three-dimensional In2O3 nanoparticles under heat treatment. Three typical samples collected from the same process were subsequently applied as electrocatalysts for the CO2 reduction reaction (CO2RR). Benefiting from the abundant oxygen defects, a faradaic efficiency of 93.5% toward formate at moderate overpotentials (−0.9 V vs. RHE) with a partial current density over 14 mA cm−2 was obtained over Vo-rich InOOH nanosheets. This research provides a strategy to design a highly efficient system for the CO2RR and sheds new light on the structure–activity relationship of low-dimensional oxidized metal catalysts.


 Please wait while we load your content...
Please wait while we load your content...